A beachhead on the island of stability
DOI: 10.1063/PT.3.2880
Remember learning the periodic table of elements in high school? Our chemistry teachers explained that the chemical properties of elements come from the electronic shell structure of atoms. Our physics teachers enriched that picture of the atomic world by introducing us to isotopes and the Segrè chart of nuclides, which arranges them by proton number Z and neutron number N.
Nuclear binding energies and nuclear shell structures provide organizing principles for understanding the properties of nuclei and the directions of radioactive decay. Protons and neutrons occupy a range of nuclear states with unevenly spread energies. Groups of those states with relatively close energies form so-called shells separated by large energy gaps, analogous to electron shells in atomic physics.
At certain numbers of protons and neutrons, called magic numbers, nuclear shell closures occur, and consequently, a large amount of energy is needed to excite a nucleon to the distant higher level. Closed nuclear shells enhance nuclear stability. (See the article by David Dean, Physics Today, November 2007, page 48
Despite much recent progress, scientists are still learning how to consistently describe the structure and properties of atoms and nuclei. Experimental studies of the heaviest elements and nuclei continue to yield new data that challenge and inform theoretical models of atomic and nuclear structures.
The overarching questions are far from trivial. Where is the end of the periodic table? What is the heaviest nucleus? How do properties evolve for extreme numbers of protons, neutrons, and electrons? How do relativistic electrons in outer atomic orbits influence the size and chemical behavior of the heaviest atoms? Do superheavy elements (SHEs), say those beyond Z = 100, follow the well-established group structure of the periodic table, or do they exhibit unexpected chemical and nuclear properties?
In the 1960s, developments by Vilen Strutinsky 1 and others in the understanding of the nuclear structure of lighter nuclei led to predictions by Adam Sobiczewski and his collaborators on new nuclear shell closures. 2 At about the same time, two groups—William Myers and Władysław Świątecki, and Victor Viola Jr and Glenn Seaborg—independently predicted the existence of heavy nuclei that would occupy a so-called island of stability on the Segrè chart. 3 Since that time, the concept of an island of stability has dominated the physics of superheavy nuclei.
Figure 1 shows the grand nuclear landscape as it is understood today. A handful of magic numbers have been well established: 2, 8, 20, 28, 50, and 126 (neutrons only). Modern theoretical approaches, supported by new experimental data, also point to the existence of long-lived nuclei at and around the magic neutron number N = 184. Presently, 177 is the largest experimentally observed neutron number
4
,
5
—in isotopes 294117 and livermorium-293 (

Figure 1. The grand nuclear landscape. Nuclei that have been experimentally identified are inside the yellow region, whereas nuclei only predicted to exist are roughly indicated by the green area. Black squares mark stable isotopes. Magic proton and neutron numbers, at which nuclei have enhanced stability, are indicated by red lines. The star labeled SHE indicates the region of superheavy elements. (Courtesy of Witold Nazarewicz).
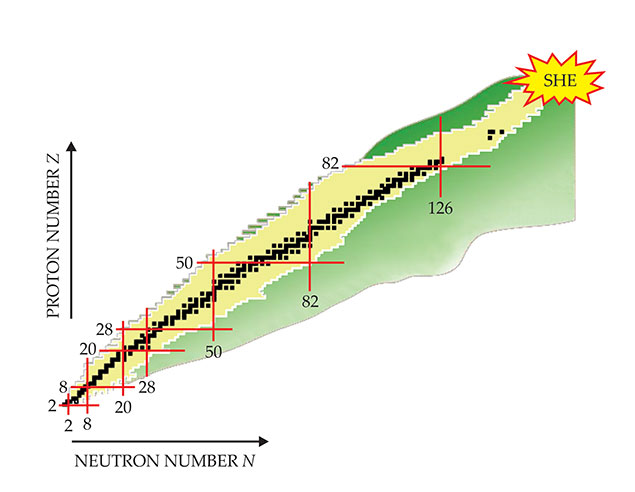
Although there is no consensus on the next magic atomic number Z, theoretical predictions place it between 114 and 126. In fact, in the vicinity of N = 184 and Z = 114 to 126, with very high neutron and proton numbers and large densities of neutron and proton states, the stability of nuclei might be enhanced without sizeable energy gaps. 6
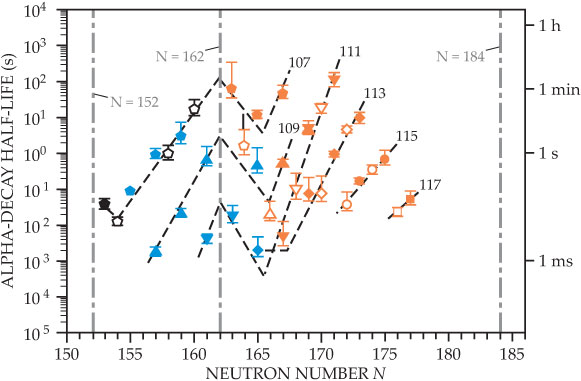
Current experimental results are consistent with the existence of an extended island of superheavy nuclei that are more resistant to radioactive decay and have much longer half-lives than somewhat lighter isotopes, as shown in figure 2. Indeed, some models predict half-lives up to a million years for new superheavy nuclei, and some even calculate them at around Earth’s age.

Figure 2. The dependence of alpha decay half-lives on neutron number. Solid and open symbols correspond to nuclei with odd and even numbers of neutrons, respectively. The symbols indicate atomic numbers—pentagons for 107, triangles for 109, upside-down triangles for 111, diamonds for 113, circles for 115, and squares for 117. The dashed lines are guides to the eye and labeled with corresponding atomic numbers. Nuclei synthesized in hot-fusion reactions with calcium-48 projectiles are shown in orange; in cold-fusion reactions, blue; and in other reactions, black. For nuclei with neutron number N > 165, half-lives grow by orders of magnitude with increasing N. The stability of those nuclei against alpha decay is enhanced for larger neutron numbers. The positions of neutron magic numbers, the established N = 152 and N = 162 and the predicted N = 184, are indicated by gray dot-dashed vertical lines.
Extensive chemistry studies have searched for traces of SHEs in geothermal waters, rare ores, and cosmic rays. However, to date all known elements beyond uranium (Z = 92) have been manmade first, in laboratories using large-scale devices like nuclear reactors and particle accelerators or in the violent conditions of nuclear explosions.
Fusion evaporation
Since the discovery of nuclear fission in 1938 (see the article by Michael Pearson, Physics Today, June 2015, page 40
The dreams of medieval alchemists have nearly come true in the modern era of accelerators and nuclear reactions.
7
For example, one can fuse two metallic atoms, a germanium-74 (
That initial
In the
The beta decay of
The development of powerful heavy-ion accelerators, from the 1970s on, gave a significant boost to experiments aiming to create new elements. By using
In particular, elements 107–112 were discovered at the separator for heavy-ion products (SHIP) facility at the GSI Helmholtz Centre for Heavy Ion Research in Germany. The beam energy in those experiments was kept low to create compound nuclei, such as
Each new step down the periodic table required greater projectile charge and mass, higher beam intensity, and longer irradiation time to fuse the colliding nuclei. The probability for creating fusion-evaporation residues, measured in terms of the cross section for each nuclear reaction channel, drops exponentially with increasing projectile atomic number.
The last and most challenging cold-fusion experiment, which used a
In the late 1990s, researchers at the Joint Institute for Nuclear Research (JINR) in Russia successfully applied a new method to create the heaviest elements and nuclei to date.
5
Instead of stable target nuclei, like
Improvements in ion-source technology enabled high beam intensities, up to 1013 projectiles per second on target, at low Ca consumption, about 0.5 mg/h, during many months of continuous target irradiation (see figure 3a). The compound nuclei created by the fusion of actinide target nuclei and

Figure 3. Superheavy elements require heavyweight facilities. (a) The heavy-ion cyclotron U 400 at the Joint Institute for Nuclear Research in Russia is among the few accelerators in the world capable of providing intense beams of calcium-48 projectiles for research on superheavy nuclei. (Courtesy of the Joint Institute for Nuclear Research.) (b) Radioactive actinide target materials are made at the High Flux Isotope Reactor at Oak Ridge National Laboratory in the US. (c) At the Radiochemical Engineering Development Center next door to the reactor, the actinides are chemically separated from irradiated pellets inside heavily shielded hot cells. Program associate Karen Murphy is shown at a hot-cell window, working with manipulators inside. (d) Shown here is 22 mg of highly purified berkelium-249, the target material used for the discovery of element 117. (Courtesy of Oak Ridge National Laboratory.)

The cross section for the synthesis of isotope 293117 in the hot-fusion reaction between a
The JINR experiments used a series of actinide targets to create elements 113–118 and their isotopes. The manmade target materials, having atomic numbers between Z = 93 (neptunium) and Z = 98 (Cf), were made at Oak Ridge National Laboratory (ORNL) in the US (see figures 3b–d) and at the Research Institute of Atomic Reactors in Russia.
At ORNL, the High Flux Isotope Reactor can produce the heaviest isotopes of the transuranium elements in quantities of tens to hundreds of milligrams by neutron irradiation of americium (Am, Z = 95) and curium (Cm, Z = 96) isotopes.
13
The Radiochemical Engineering Development Center next door separates individual elements from the irradiated Am and Cm seed material. The production and purification of
Ion separators
When heavy-ion projectiles smash into a target, various reaction products, of which only a few are fusion-evaporation residues, are ejected. All those particles, and the primary beam, enter an ion separator that uses a combination of magnetic and electric fields to select ions of interest and guide them to detectors.
The intensity of heavy-ion beams used for the production of superheavy nuclei is on the order of 1013 projectiles per second. In contrast, the rate of SHE events produced at the target varies from a single event every few days to a single event in several months. Obviously, the main purposes of a separator designed for SHE studies are to efficiently transmit SHE nuclei that are only rarely produced in the target and to reject the massive amount of primary-beam particles and other unwanted reaction products that can cause false signals.
The two most successful separators are the GSI’s SHIP, in operation since the late 1970s, and the Dubna gas-filled recoil separator (DGFRS), in service since the late 1990s at the JINR. Six new elements, from bohrium (Bh, Z = 107) to Cn (Z = 112), were discovered at SHIP, and six new elements from Z = 113 to Z = 118 were first detected at the DGFRS.
To pick out the evaporation residues from the primary beam and other reaction products, SHIP operates in vacuum with a velocity filter, which takes advantage of the different velocities of recoiling nuclei entering the separator. Because the residues receive all their momenta from the projectiles in the primary beam but are four to six times as heavy as those projectiles, they move more slowly. In the velocity filter, which is made of two electric deflectors and several dipole and quadrupole magnets, the ratio of electric and magnetic fields can be set to allow only particles with a particular velocity to pass through. Typical transmission efficiency at SHIP for fusion-evaporation residues is about 30%, and the primary beam suppression factor is more than 1010.
Unwanted particles—such as transfer-reaction products produced when a target nucleus absorbs one or more nucleons from a projectile, light particles emitted from the target, and scattered beam particles—still reach the detectors at a rate of a few hundred per second, compared with about one event per week (about 2 × 10−6/s) for SHE evaporation residues. Other recoil mass separators have been built following the same principle as SHIP—for example at ORNL and Argonne National Laboratory in the US, at the National Large Heavy Ion Accelerator in France, and at the JINR.
The DGFRS utilizes a different concept for transmitting superheavy recoils from the target area and rejecting primary beam particles. The separator is filled with hydrogen at a pressure of about 1 torr and consists of a 23° dipole bending magnet and a pair of quadrupole magnets (see figure 4). All recoiling fusion-evaporation residues have their ionic charges reset to an average value through collisions with hydrogen atoms. The transmission efficiencies—typically about 35% for the products of a

Figure 4. The Dubna gas-filled recoil separator is outfitted with a dipole bending magnet (D) and two ion-focusing quadrupole magnets (Q) to select and guide the superheavy recoils (red) from collisions between calcium-48 projectiles (blue) and a rotating actinide target to a set of detectors. The inset shows the detector station with two time-of- flight detectors and silicon-stack detectors. (Adapted from Y. T. Oganessian et al., Phys. Rev. C 83, 054315, 2011,
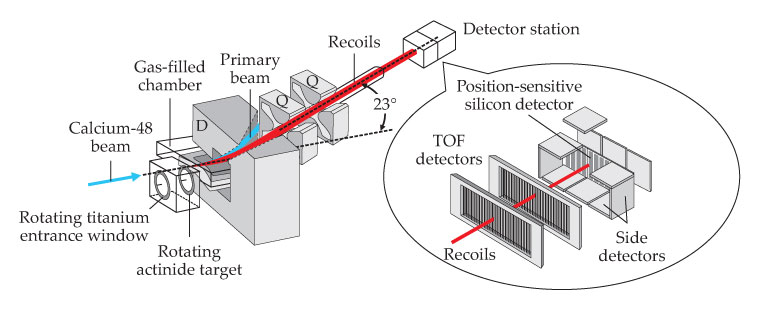
The detection of superheavy nuclei is similar at SHIP and the DGFRS. The nuclei selected by the electromagnetic fields and transmitted to the focal plane of the separator pass through time-of-flight (TOF) detectors that give timing signals. For the DGFRS, they are two multiwire proportional counters filled with 1.7 torr of pentane gas and operated at 1500 V. The combined efficiency of the TOF counters is about 99.9%.
The ions are shallowly implanted into an approximately 5000-mm2 position-sensitive silicon counter that is surrounded by several Si side detectors. When an alpha emission indicates an implanted radioactive superheavy nucleus, the alpha energy and the position and time of implantation are recorded. Side detectors catch about 37% of the alpha particles emitted at backward angles, whereas those going forward are fully absorbed in the implantation detector with nearly 100% efficiency. The transactinide separator and chemistry apparatus at the GSI, the Berkeley gas-filled separator at LBNL, and the gas-filled recoil ion separator at RIKEN are similar spectrometers used for SHE studies.
Spotting the heaviest nuclei
The fusion of a projectile ion and a target actinide atom into a superheavy nucleus in its ground state is an extremely rare event. To identify such an event and distinguish it from the background of scattered multinucleon transfer products, researchers rely on the measured characteristics of signals generated by the recoiling evaporation residue itself and of its chain of several sequential decays.
The superheavy nucleus triggers the TOF detectors and creates a valid TOF signal. Then it becomes lodged in the implantation counter and, for gas-filled separators, deposits around 8–15 MeV of energy. The implantation signal is followed seconds or even milliseconds later by the detection of an alpha particle at the same position, with a resolution of a few square millimeters down to 1 mm2.
If the alpha particle’s energy is close to the expected one for a superheavy nucleus, the signal triggers a beam stop to block the primary beam. That way, subsequent decays are observed without beam-associated background. When another alpha decay is observed at the same position, the beam pause is prolonged, sometimes to several minutes or even many hours, to detect the final decay—a spontaneous fission signal. Such low-background operation mode causes losses of about 5–10% in real experiment time as a tradeoff for significant gain in sensitivity to rare events, in particular for longer-lived daughter nuclei.
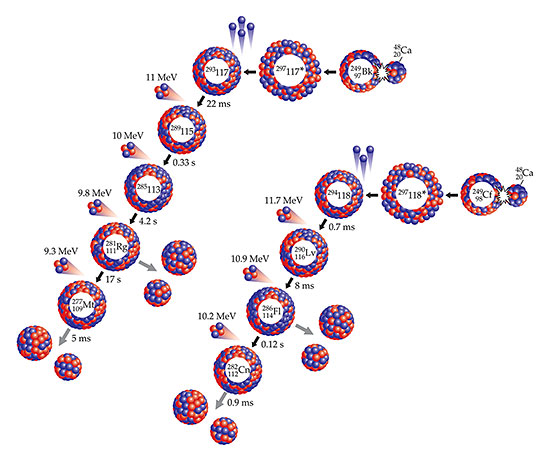
So far, all decay chains of nuclei at the so-called hot-fusion island, the region of superheavy nuclei created using actinide targets and

Figure 5. Decay chains for isotopes 293117 and 294118, showing the half-life and alpha-decay energy of each nucleus in the chains. Black arrows indicate alpha decay and gray arrows indicate spontaneous fission. In both cases, hot-fusion reactions between calcium-48 projectiles and actinide target materials, either berkelium-249 or californium-249, produce compound nuclei, labeled with asterisks, that promptly evaporate off several neutrons. Toward the ends of the chains, roentgenium-281 (from 293117) and flerovium-286 (from 294118) can spontaneously fission or alpha decay into meitnerium-277 and copernicium-282, respectively. Both end-chain nuclei undergo spontaneous fission.
The probability for a random sequence of events to simulate a long decay chain is extremely small, 14 usually at the level of 10−6 to 10−12. Such a sequence starts with a nucleus triggering a valid TOF signal, followed by implantation at the right energy and a chain of alpha-like signals terminating in spontaneous fission, all occurring at the same place and within a relatively limited time frame.
Decay studies involving superheavy ion implantation and alpha spectroscopy allow researchers to deduce only the half-life and energy of the emitted particle. Decay chains originating from some odd-mass nuclei and from odd-Z and odd-N isotopes display richer spectra of alpha energies, which indicate the population of low-energy excited states in daughter nuclei.
Recent studies of decay chains that start from 288115 nuclei complemented alpha spectroscopy with gamma- and x-ray detectors.
11
The experiments detected photons emitted from excited states of the daughter nuclei meitnerium-276 (
Nuclei with both even Z and even N at the island of stability are characterized by relatively large rates of fission and of alpha decay. The daughter nuclei, after alpha decay, also have even proton and neutron numbers. In such nuclei, all the protons and neutrons are paired, so the ground states have spin 0 and even parity.
The alpha decay occurs with orbital angular momentum l = 0, which means that the alpha particle has to tunnel through only a coulomb barrier and not an additional orbital angular momentum barrier. That scenario results simultaneously in fast alpha decay and high fission probability. Consequently, we observe relatively short decay chains that end in spontaneous fission in even–even nuclei. Fission probabilities are about 1000 times lower for odd-mass nuclei and more limited for those with both odd Z and odd N, so they have longer alpha-decay chains. However, they also end in fission of the terminating nuclei.
Discovery and exploration
During the past 15 years, more than 50 superheavy nuclei and six new elements, from Z = 113 to Z = 118, have been synthesized in hot-fusion reactions between
All decay chains observed at the hot-fusion island end in spontaneous fission and do not connect to the nuclear mainland. In 2013 the alpha decay of
The currently accepted assignments of mass and atomic numbers for nuclei at the hot-fusion island are the only consistent ones possible. The nuclei are connected by decay chains, and the assignments are cross checked by producing the involved nuclei in different nuclear reactions, a procedure called cross bombardment.
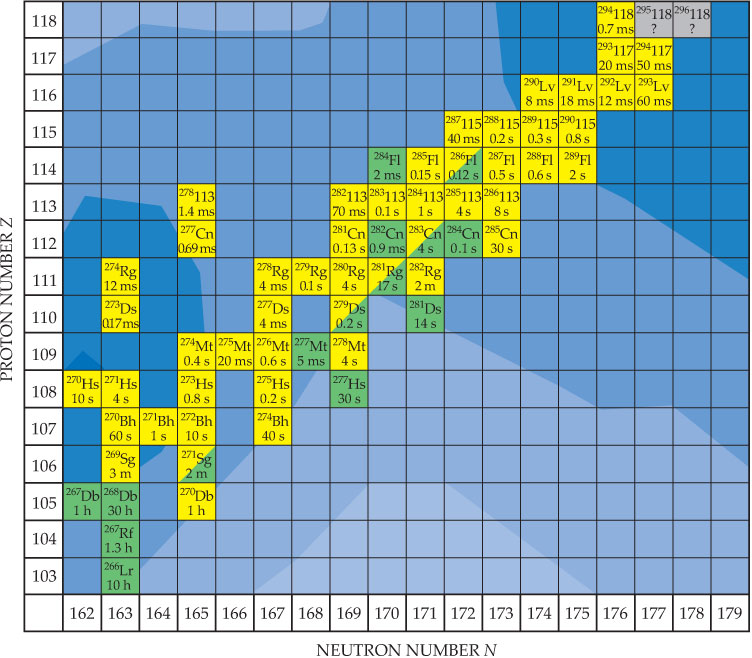
In addition, the measured excitation functions, the beam-energy dependence of the cross sections for different reaction channels, offer a consistent picture of identified nuclei synthesized in different laboratories. The new nuclei synthesized at the island of stability are presented in figure 6.

Figure 6. The top of the nuclear landscape, with the heaviest identified nuclei and their half-lives and decay modes—yellow for alpha decay and green for spontaneous fission. The contoured color background indicates the predicted stability of nuclei—the darker the color, the more stable the nucleus. A Russia–US team is currently focusing on new isotopes 295118 and 296118 (gray squares) using a calcium-48 beam and an isotopically mixed californium target. (Background contour plot courtesy of the GSI Helmholtz Centre for Heavy Ion Research.)
In addition to developing improvements in decay spectroscopy of nuclei, several groups are attempting to directly measure the mass numbers of nuclei at the hot-fusion island. Nobelium-252 (
The quest continues for new elements and nuclei closer to the next magic neutron number predicted at N = 184. Recently, researchers at ORNL produced a target containing a mix of
An expansion of the periodic table beyond element 118 will require projectiles with atomic numbers greater than 20 that are heavier than
New techniques in the physics of superheavy nuclei include next-generation accelerator facilities like the Superheavy Element Factory under construction at the JINR, new technologies for the production of robust targets, and the creation of sophisticated detector arrays. Such developments should allow the production of the heaviest nuclei in larger quantities and advance our understanding of the fundamental properties of nuclear structure and atomic properties at the top of nuclear and atomic worlds.

WINSLOW HOMER

References
1. V. M. Strutinsky, Nucl. Phys. A 95, 420 (1967); https://doi.org/10.1016/0375-9474(67)90510-6
122, 1 (1968). https://doi.org/10.1016/0375-9474(68)90699-42. A. Sobiczewski, F. A. Gareev, B. N. Kalinkin, Phys. Lett. 22, 500 (1966). https://doi.org/10.1016/0031-9163(66)91243-1
3. W. D. Myers, W. Świątecki, Nucl. Phys. 81, 1 (1966); https://doi.org/10.1016/S0029-5582(66)80001-9
V. E. Viola Jr, G. T. Seaborg, J. Inorg. Nucl. Chem. 28, 741 (1966). https://doi.org/10.1016/0022-1902(66)80412-84. Y. T. Oganessian et al., Phys. Rev. Lett. 104, 142502 (2010). https://doi.org/10.1103/PhysRevLett.104.142502
5. Y. T. Oganessian, J. Phys. G 34, R165 (2007). https://doi.org/10.1088/0954-3899/34/4/R01
6. M. Bender, W. Nazarewicz, P.-G. Reinhard, Phys. Lett. B 515, 42 (2001). https://doi.org/10.1016/S0370-2693(01)00863-2
7. G. Münzenberg, M. Schädel, Moderne Alchemie: Die Jagd nach den schwersten Elementen, Vieweg (1996).
8. K. Morita et al., J. Phys. Soc. Jpn. 81, 103201 (2012). https://doi.org/10.1143/JPSJ.81.103201
9. Y. T. Oganessian et al., Phys. Rev. C 87, 054621 (2013). https://doi.org/10.1103/PhysRevC.87.054621
10. C. E. Düllmann et al., Phys. Rev. Lett. 104, 252701 (2010); https://doi.org/10.1103/PhysRevLett.104.252701
S. Hofmann et al., Eur. Phys. J. A 48, 62 (2012); https://doi.org/10.1140/epja/i2012-12062-1
J. Khuyagbaatar et al., Phys. Rev. Lett. 112, 172501 (2014). https://doi.org/10.1103/PhysRevLett.112.17250111. D. Rudolph et al., Phys. Rev. Lett. 111, 112502 (2013). https://doi.org/10.1103/PhysRevLett.111.112502
12. L. Stavsetra et al., Phys. Rev. Lett. 104, 132502 (2010); https://doi.org/10.1103/PhysRevLett.104.132502
K. Morita et al., in RIKEN Accelerator Progress Report 2013, vol. 47, RIKEN Nishina Center for Accelerator-Based Science (2014), p. xi.13. J. B. Roberto et al., in Fission and Properties of Neutron-Rich Nuclei: Proceedings of the Fifth International Conference on ICFN5, Sanibel Island, Florida, USA, 4–10 November 2012, J. H. Hamilton, A. V. Ramayya, eds., World Scientific (2014), p. 287.
14. K.-H. Schmidt et al., Z. Phys. A 316, 19 (1984). https://doi.org/10.1007/BF01415656
15. E. Minaya Ramirez et al., Science 337, 1207 (2012). https://doi.org/10.1126/science.1225636
16. A. M. Rodin et al., Instrum. Exp. Tech. 57, 386 (2014). https://doi.org/10.1134/S0020441214030208
17. N. Esker et al., Bull. Am. Phys. Soc. 59(10), 54 (2014).
18. Y. T. Oganessian et al., Phys. Rev. C 79, 024603 (2009); https://doi.org/10.1103/PhysRevC.79.024603
S. Hofmann et al., in GSI Scientific Report 2008, K. Grosse, ed., GSI Report 2009-1, GSI Helmholtz Centre for Heavy Ion Research (2009), p. 131.
More about the authors
Yuri Ts. Oganessian is the scientific leader of the Flerov Laboratory of Nuclear Reactions at the Joint Institute for Nuclear Research in Dubna, Russia. Krzysztof P. Rykaczewski is a senior researcher at Oak Ridge National Laboratory in Oak Ridge, Tennessee.






